Use code RJTJFG for 20% off at checkout. Until 5/31/20



The watermark at the lower right corner of the image will not appear on the final product.

by NIST/Science Source
$34.00
Color

Image Size
Product Details
Our youth t-shirts are made from 100% pre-shrunk cotton and are available in five different sizes. All youth t-shirts are machine washable.
Design Details
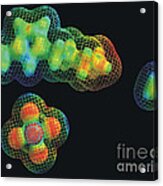
Molecular space filling models demonstrate the difference in size for the positively charged cation (top image) and the negatively charged anion... more
Ships Within
1 - 2 business days

Photograph

Canvas Print

Framed Print

Art Print

Poster

Metal Print
Acrylic Print

Wood Print

Greeting Card

iPhone Case

Throw Pillow

Duvet Cover

Shower Curtain

Tote Bag

Round Beach Towel

Zip Pouch

Beach Towel

Weekender Tote Bag

Portable Battery Charger

Bath Towel

Apparel

Coffee Mug

Yoga Mat

Spiral Notebook

Fleece Blanket

Tapestry

Jigsaw Puzzle

Sticker
Molecular "space filling" models demonstrate the difference in size for the positively charged "cation" (top image) and the negatively charged "anion" (bottom left) that combine to form a promising ionic liquid. It is still a mystery how the much smaller water molecule (right) can have such a large effect on the viscosity of such ionic liquids. People in industry are very interested in using ionic liquids because unlike most organic solvents, they don't evaporate and they are not flammable. A cation is an ion with fewer electrons than protons, giving it a positive charge. An anion is an ion with more electrons than protons, giving it a net negative charge. An ionic liquid (IL) is a salt in the liquid state. While ordinary liquids such as water and gasoline are predominantly made of electrically neutral molecules, ionic liquids are largely made of ions and short-lived ion pairs. These substances are variously called liquid electrolytes, ionic melts, ionic fluids, fused salts, liquid sal...
$34.00


There are no comments for Ionic Liquid. Click here to post the first comment.